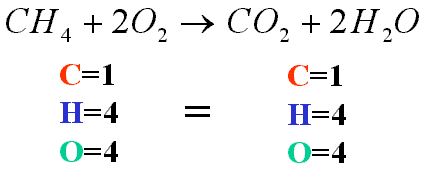Balancing equations
Types of chemical reactions
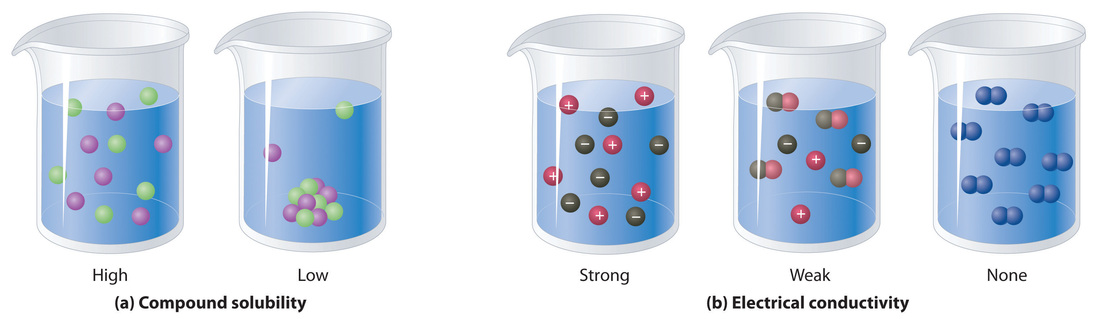
Ions dissolved in water
- The ions break into their contituent radical ions.
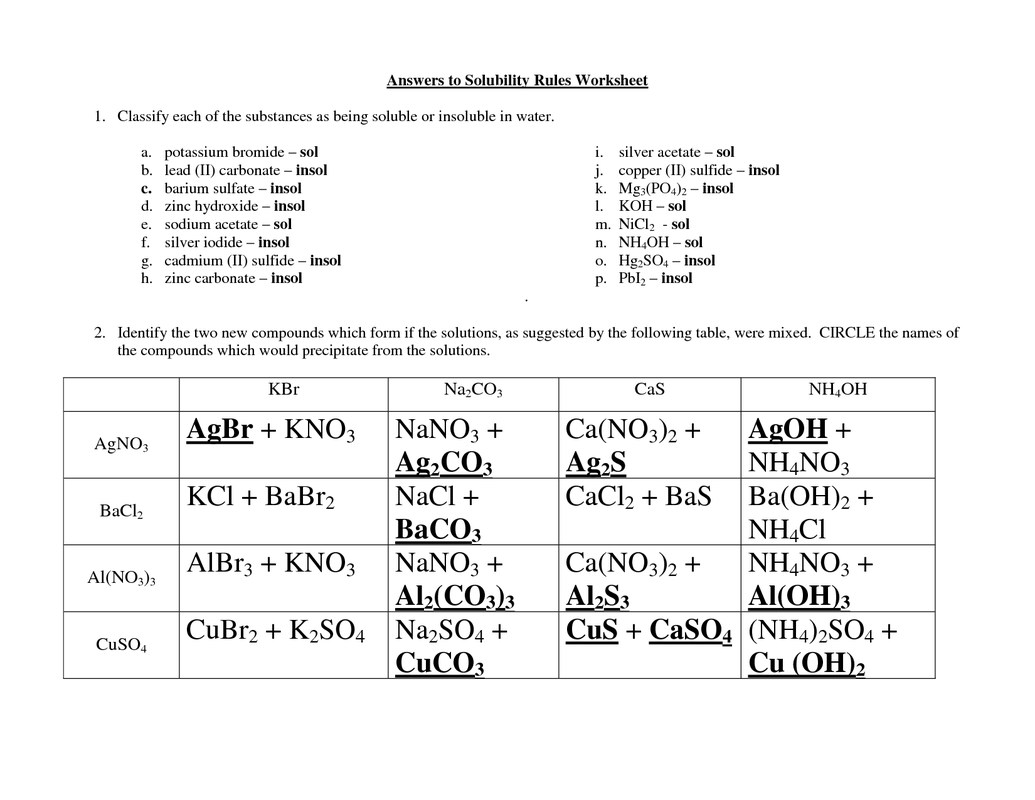
Solubility rules
Click on these links to take you to websites and videos where you can get extra help on these topics
Activities:
Chapter 7 vocabulary quizlet
http://quizlet.com/32097349/chemistry-chapter-67-flash-cards/
Types of reactions quizlet
http://quizlet.com/31739557/types-of-reactions-flash-cards/
Balancing chemical equations activity
http://www.sciencegeek.net/Chemistry/taters/EquationBalancing.htm
Solubility rules quizlet
http://quizlet.com/11343860/solubility-flash-cards/
Balancing Chemical Equations:
Video- http://www.youtube.com/watch?v=RnGu3xO2h74
http://education.jlab.org/elementbalancing/
http://funbasedlearning.com/chemistry/chemBalancer/default.htm
*This activity helps with balancing equations and is a fun way to learn balancing.
Solubility Rules:
Video: http://www.youtube.com/watch?v=AsCLuLS-yZY
http://www.csudh.edu/oliver/chemdata/solrules.htm
Types of Chemical Reactions:
Video: http://www.youtube.com/watch?v=a7PZDEeqjcU
http://misterguch.brinkster.net/6typesofchemicalrxn.html
Solubility:
Video: http://www.youtube.com/watch?v=zoDrlrTVLOU
http://www.chem.wisc.edu/deptfiles/genchem/sstutorial/Text11/Tx112/tx112.html
http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch18/soluble.php
*These websites provide examples and activities for both insoluble and soluble solids.
Ionic Compounds dissolving in water
http://www.youtube.com/watch?v=Vgh_uB6QUBo
*This video explains how an ionic compound dissolves in water and what are the characteristics of an ionic compound that dissolves in water.
Soluble/Insoluble solids
http://www.youtube.com/watch?v=ek6CVVJk4OQ
*This is a video that explains what makes a solid soluble or insoluble.
Solubility Rules
http://www.youtube.com/watch?v=5vZE9K9VaJI
*This is a video explaining the solubility rules and how to use them.
Predicting Products
http://www.youtube.com/watch?v=P0jG2TjLyGI
*This video helps explain how to predict the products for chemical reactions.
Balancing Chemical Equations
http://www.youtube.com/watch?v=ZzwziP04448
*This video explains how to balance equations, and this video also shows and gives examples of how to balance chemical equations.
- Reactants ----------> Product
- (s)- solid
- (l)- liquid
- (g)-gas
- (aq)- aqueous solution
Types of chemical reactions
- 1. Combustion.
- CH4+O2 -> CO2 + H2O
- Always look for in the reactant a carbon source and oxygen gas and in the product CO2
- CH4+O2 -> CO2 + H2O
- Synthesis
- Adding elements and/or compounds to form a new single compound
- example: H2 +O2-> H2O
- Decomposition
- Breaking a compound into simpler parts
- Example: H2O-> H2 +O2
- Single displacement
- One element/compound replaces another one.
- note only a cation can replace a cation and an anion can only replace an anion
- example: Zn +FeCl2-> Fe + ZnCl2
- Double displacement
- Elements/compounds switches partner to form 2 new compound.
- Example: AgNO3 (aq)+ NaCl(aq) -> AgCl(s) + NaNO3(aq)
- Acid/ base reaction
- In the reactant, there must be both an acid(H) and a base(OH).
- example: HNO3 + NaOH->H2O + NaNO3
- Precipitation reaction:
- Formation of a solid when two or more aqueous solution are combined
- AgNO3 (aq)+ NaCl(aq) -> AgCl(s) + NaNO3(aq)
Ions dissolved in water
- The ions break into their contituent radical ions.
Solubility rules
- 1. Most nitrate( NO3 -1) are soluble
- 2. Most salts of Na+,K+ and NH4+ are soluble
- 3. Most chloride salts are soluble.
- exception: Ag+,Pb2+ , Hg2 +2
- 4. Most sulfates salts are soluble
- exception:Ba2+, Pb2+ , Ca2+
- 5. Most hydroxide (OH-)compounds are insoluble
- Exception- ions in rule number 2 and Ba/Ca
- 6.Most sulfides, carbonate, phosphate are insoluble
- Exception- ions in rule number 2
- All simple Decomposition Reactions begin with: compound →
- In the example: H2O →
- break the compound into its individual elements, H and O
- H + O
- When elements are ALONE in an equation, ask yourself "are they one of the 7 diatomic molecules?"
- In this case, both of the products ARE diatomic molecules.
- The correct chemical equation is: H2O → H2 + O2
All simple Synthesis Reactions begin with: element + element →- In the example: O2 + Al →
- Combine the elements to make a compound.
- Write the metal (element with a positive oxidation number) first and the nonmetal (element with a negative oxidation number second.
- AlO
- Now use subscripts to make the oxidation numbers total zero. (aluminum is +3 and oxygen is −2)
- The correct chemical equation is: O2 + Al → Al2O3
All simple Single Displacement Reactions begin with: element + compound →- In the example: Zn + HCl →
- Zinc (with a positive oxidation number) will switch places with the positive part of the compound.
- H + ZnCl
- Is hydrogen one of the diatomic molecules?
- (yes, it must have a subscript of 2) H2 + ZnCl
- Use subscripts to make the oxidation numbers in the compound total zero. (Zn is +2 and Cl is −1)
- The correct chemical equation is: Zn + HCl → H2 + ZnCl2
All simple Double Displacement Reactions begin with: compound + compound →- In the example: FeCl3 + NH4OH →
- Switch the positive parts of the two compounds.
- NH4Cl + FeOH
- Use subscripts to make the oxidation numbers in both compounds total zero.
- The polyatomic ion NH4 is +1
- Chlorine is −1
- Fe is +3 on the left and you can assume it will remain +3 on the right
- The polyatomic ion OH is −1
- The correct chemical equation is: FeCl3 + NH4OH → NH4Cl + Fe(OH)3
All simple Combustion Reactions begin with: hydrocarbon + oxygen → - In the example: C6H6 + O2 →
- The products of combustion are ALWAYS carbon dioxide and water.
- The correct chemical equation is: C6H6 + O2 → CO2 + H2O
- 4 common “driving forces” for reaction
- Formation of a solid(ex precipitation)
- Formation of water(ex. combustion)
- Transfer of electron( ex. oxidation- reduction)
- Formation of gas(ex. combustion)
- 1. Molecular equation
- No charges are showing. Only compounds
- AgNO3 (aq)+ NaCl(aq) -> AgCl(s) + NaNO3(aq)
- II. Complete ionic equations
- All (aq) compounds will disassociate “break” into their ions
- Ag+(aq) +NO3- (aq)+ Na+(aq) +Cl-(aq) -> AgCl(s) + Na+ (aq) +NO3-(aq)
- III. Net ionic equation
- Find the solid and then write the ions in the reactants that makes up the product
- Ag+(aq) + Cl-(aq)-> AgCl(s)
Click on these links to take you to websites and videos where you can get extra help on these topics
Activities:
Chapter 7 vocabulary quizlet
http://quizlet.com/32097349/chemistry-chapter-67-flash-cards/
Types of reactions quizlet
http://quizlet.com/31739557/types-of-reactions-flash-cards/
Balancing chemical equations activity
http://www.sciencegeek.net/Chemistry/taters/EquationBalancing.htm
Solubility rules quizlet
http://quizlet.com/11343860/solubility-flash-cards/
Balancing Chemical Equations:
Video- http://www.youtube.com/watch?v=RnGu3xO2h74
http://education.jlab.org/elementbalancing/
http://funbasedlearning.com/chemistry/chemBalancer/default.htm
*This activity helps with balancing equations and is a fun way to learn balancing.
Solubility Rules:
Video: http://www.youtube.com/watch?v=AsCLuLS-yZY
http://www.csudh.edu/oliver/chemdata/solrules.htm
Types of Chemical Reactions:
Video: http://www.youtube.com/watch?v=a7PZDEeqjcU
http://misterguch.brinkster.net/6typesofchemicalrxn.html
Solubility:
Video: http://www.youtube.com/watch?v=zoDrlrTVLOU
http://www.chem.wisc.edu/deptfiles/genchem/sstutorial/Text11/Tx112/tx112.html
http://chemed.chem.purdue.edu/genchem/topicreview/bp/ch18/soluble.php
*These websites provide examples and activities for both insoluble and soluble solids.
Ionic Compounds dissolving in water
http://www.youtube.com/watch?v=Vgh_uB6QUBo
*This video explains how an ionic compound dissolves in water and what are the characteristics of an ionic compound that dissolves in water.
Soluble/Insoluble solids
http://www.youtube.com/watch?v=ek6CVVJk4OQ
*This is a video that explains what makes a solid soluble or insoluble.
Solubility Rules
http://www.youtube.com/watch?v=5vZE9K9VaJI
*This is a video explaining the solubility rules and how to use them.
Predicting Products
http://www.youtube.com/watch?v=P0jG2TjLyGI
*This video helps explain how to predict the products for chemical reactions.
Balancing Chemical Equations
http://www.youtube.com/watch?v=ZzwziP04448
*This video explains how to balance equations, and this video also shows and gives examples of how to balance chemical equations.
This example represents the differences between solids that dissolve in water and substances that do not dissolve in water.
This table is an example of the solubility rules needed to determine if a compound dissolves in water or not.
Solubility
Solubility
- Is this compound soluble or insoluble based on the solubility rules?
- AgNO3
- The compound is soluble because nitrates are soluble.
- AgCl
- The compound is insoluble because Chlorides are soluble with the exceptions of Ag, Hg, Pb.
This is an example of the solubility rules and using them to determine whether or not a compound is soluble or insoluble, and this can also be used to predict products dependent on solubility.
This is an example of how to predict the products for a chemical reaction.
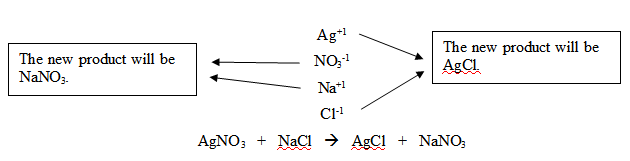
Predicting the Products
- AgNO3 (aq)+ NaCl(aq) ->
- find the products for this this equation
- AgNO3 (aq)+ NaCl(aq) -> AgCl + NaNO3
This shows how to predict the products for a reaction and also how you pair them to make new compounds.
This is an example of balancing chemical equations.