Chapter 5: Nomenclature
Naming Compounds
-a compound can contain a metal and a nonmetal
-a compound can also contain 2 nonmetals
Binary Compounds
-they contain a positive ion (cation) and a negative ion(anion)
Ionic Compounds
-type I: the metal will only have one charge
-type II: the metal may form more than one cation causing it to have more than one charge
-deal more with the transition metals(can have more than one charge)
-have systematic names that relate to the charge of the metal
-type III:these compounds can contain 2 or more nonmetals that are covalently bonded
Rule for naming type I ionic compounds:
1. Cation is always named first followed by the anion
Cation has the same name as the element and the anion will have the ending "-ide"
Rule for naming type II ionic compounds:
1. Metal name(roman numeral) anion name
example: Iron(II) Oxide
Rules for naming type III compounds
1. first element in the formula is named first and the full element name is used
2. second element is named as an anion
3. prefixes are used to show how many there are of each element.
-never put "mono" if the first element only has 1
-example: CO is Carbon monoxide not "Mono"carbon monoxide
Rules for naming acids:
* Acid will have an H (H=Cation)
1. must determine the name of the anion before you can name the acid
2. know it will be an acid because i has H in front
3. if oxygen is not present in the compound must have hydro- in front of the name
4. -ate and -ide change to -ic
5. -ite endings change to -ous
Sample: HClO2
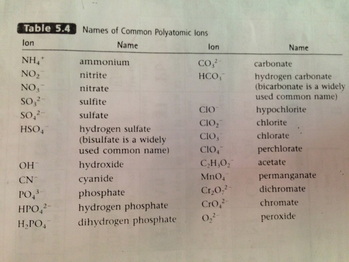
Polyatomic Ions:
1. these ions contain more than 1 atom
ex: SO4^-2
2. type I or type II can be used for Polyatomics
-a compound can contain a metal and a nonmetal
-a compound can also contain 2 nonmetals
Binary Compounds
-they contain a positive ion (cation) and a negative ion(anion)
Ionic Compounds
-type I: the metal will only have one charge
-type II: the metal may form more than one cation causing it to have more than one charge
-deal more with the transition metals(can have more than one charge)
-have systematic names that relate to the charge of the metal
-type III:these compounds can contain 2 or more nonmetals that are covalently bonded
Rule for naming type I ionic compounds:
1. Cation is always named first followed by the anion
Cation has the same name as the element and the anion will have the ending "-ide"
Rule for naming type II ionic compounds:
1. Metal name(roman numeral) anion name
example: Iron(II) Oxide
Rules for naming type III compounds
1. first element in the formula is named first and the full element name is used
2. second element is named as an anion
3. prefixes are used to show how many there are of each element.
-never put "mono" if the first element only has 1
-example: CO is Carbon monoxide not "Mono"carbon monoxide
Rules for naming acids:
* Acid will have an H (H=Cation)
1. must determine the name of the anion before you can name the acid
2. know it will be an acid because i has H in front
3. if oxygen is not present in the compound must have hydro- in front of the name
4. -ate and -ide change to -ic
5. -ite endings change to -ous
Sample: HClO2
Polyatomic Ions:
1. these ions contain more than 1 atom
ex: SO4^-2
2. type I or type II can be used for Polyatomics
Calculations:
Nomenclature Type 1:
1. What is the chemical formula for this compound? Strontium Chloride
SrCl2
2. what is the Name for this compound? Rb2S
Rubidium Sulfide
3. what is the Name for this compound? Li2S
Lithium Sulfide
Nomenclature Type 2:
1. what is the Name for this compound? FeF3
Iron (III) Fluoride
2. what is the Name for this compound? CuO
Copper (II) Oxide
3. What is the chemical formula for this compound? Iron(III) Iodide
FeI3
Nomenclature Type 3:
1. What is the chemical formula for this compound? Nitrogen Triodide
NI3
2. What is the chemical formula for this compound? Sulfur Dioxide
SO2
3. what is the Name for this compound? NF3
Nitrogen trifluoride
Nomenclature Polyatomic Ions:
1. what is the chemical formula for this compound? Ammonium Hydrogen
NH4HF2
2. what is the Name for this compound? FeSO4
Iron(II) Sulfate
3. what the chemical formula for this compound? Magnesium Nitrite
Mg(NO2)2
Nomenclature Acids:
1. What is the formula of the following acid? Phosphoric Acid
H3PO4
2. What is the formula of the following acid? Hydroiodic acid
HI
3. What is the name of the following acid? HF
hydrofluoric acid
Nomenclature Type 1:
1. What is the chemical formula for this compound? Strontium Chloride
SrCl2
2. what is the Name for this compound? Rb2S
Rubidium Sulfide
3. what is the Name for this compound? Li2S
Lithium Sulfide
Nomenclature Type 2:
1. what is the Name for this compound? FeF3
Iron (III) Fluoride
2. what is the Name for this compound? CuO
Copper (II) Oxide
3. What is the chemical formula for this compound? Iron(III) Iodide
FeI3
Nomenclature Type 3:
1. What is the chemical formula for this compound? Nitrogen Triodide
NI3
2. What is the chemical formula for this compound? Sulfur Dioxide
SO2
3. what is the Name for this compound? NF3
Nitrogen trifluoride
Nomenclature Polyatomic Ions:
1. what is the chemical formula for this compound? Ammonium Hydrogen
NH4HF2
2. what is the Name for this compound? FeSO4
Iron(II) Sulfate
3. what the chemical formula for this compound? Magnesium Nitrite
Mg(NO2)2
Nomenclature Acids:
1. What is the formula of the following acid? Phosphoric Acid
H3PO4
2. What is the formula of the following acid? Hydroiodic acid
HI
3. What is the name of the following acid? HF
hydrofluoric acid
Resources:
Activity Link: This activity helps one better understand how to name compounds from chemical formulas and how to give chemical formulas from names of a compound.
Video Link: This video reinforces the rules of how to name chemical formulas correctly and goes in depth about nomenclature.
Activity Link: This activity helps one better understand how to name compounds from chemical formulas and how to give chemical formulas from names of a compound.
Video Link: This video reinforces the rules of how to name chemical formulas correctly and goes in depth about nomenclature.