Solutions
Polar/Non Polar
Aqueous Solution
Water
SOLUTION
10g sample x (5g carbon/100g sample) = 0.5g carbon
0.5g carbon x (1mol carbon/12.011g carbon) = 0.0416 mol carbon
*This is an example of a stoichiometry problem.
Solution and Concentration
Solution Composition
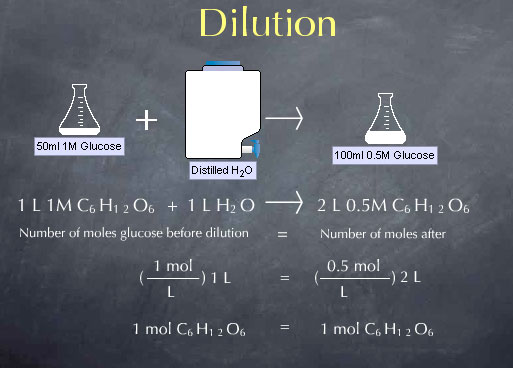
You dilute a solution whenever you add solvent to a solution.
MiVi = MfVf
where M is molarity, V is volume, and the subscripts i and f refer to the initial and final values.
Or by using the equation:
Concentration and Molarity
*Here is a calculation for the Molarity of a solution.
Step 1 Calculate the number of moles of NaOH that are in 20.0 grams.
Look up the atomic masses for the elements in NaOH from the Periodic Table. The atomic masses are found to be:
Na is 23.0
H is 1.0
O is 16.0
Plugging these values:
1 mol NaOH weighs 23.0 g + 16.0 g + 1.0 g = 40.0 g
So the number of moles in 20.0 g is:
moles NaOH = 20.0 g × 1 mol/40.0 g = 0.500 mol
Step 2 Determine the volume of solution in liters.
1 liter is 1000 cm3, so the volume of solution is: liters solution = 482 cm3 × 1 liter/1000 cm3 = 0.482 liter
Step 3 Determine the molarity of the solution.
Simply divide the number of moles by the volume of solution to get the molarity:
molarity = 0.500 mol / 0.482 liter
molarity = 1.04 mol/liter = 1.04 M
Answer
The molarity of a solution made by dissolving 20.0 g of NaOH to make a 482 cm3 solution is 1.04 M
- a homogenous mixture
- A mixture in which the compounds are uniformly intermingled
- They are mixed well so that you can’t see the parts
- 2 parts of a solution
- Solvent- substance that present in the largest amount
- Solute- substance present in a small amount when compared a substance that is present in a large amount
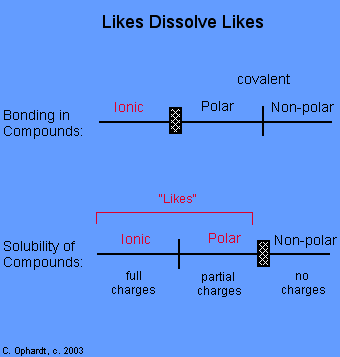
Polar/Non Polar
- Polar solvents have large dipole moments (aka “partial charges”); they contain bonds between atoms with very different electronegativities, such as oxygen and hydrogen
- Non polar solvents contain bonds between atoms with similar electronegativities, such as carbon and hydrogen. Bonds between atoms with similar electronegativities will lack partial charges; it’s this absence of charge which makes these molecules “non-polar”
- Polar solvents dissolve polar solutes.
- Nonpolar solvents dissolve nonpolar solutes.
- Polar substances and nonpolar substances generally remain separate.
Aqueous Solution
- are solutions with water as a solvent
- Water is always the solvent....
- Salt water
- NaCl is solute and water is solvent
- NaCl(s) -> NaCl(aq)
- NaCl(s) -> Na+(aq) +Cl-(aq)
- Ionic compound will dissolve in water since ionic compounds are made up of cation(+) and anion(-)
Water
- Water will also dissolve many nonionic compounds.
- Polar compound will dissolve in water
- compounds that will contain charges ( + or -)
- Not all things are soluble in water.
- Non-polar molecules will not dissolve in to polar solution.
- Example
- Polar solvents dissolve polar solutes.
- Nonpolar solvents dissolve nonpolar solutes.
- Polar substances and nonpolar substances generally remain separate.
- Stoichiometry is a section of chemistry that involves using relationships between reactants and/or products in a chemical reaction to determine desired quantitative data
SOLUTION
10g sample x (5g carbon/100g sample) = 0.5g carbon
0.5g carbon x (1mol carbon/12.011g carbon) = 0.0416 mol carbon
*This is an example of a stoichiometry problem.
Solution and Concentration
- Every soluble substances will have a limit on how much solute will go into solution.
- When this happens, the solution is called saturated.
- If you had more solute after the saturation point then it will stay solid.
- Unsaturated solution- this type of solution occurs with a solution has not reached the limit of the solute.
Solution Composition
- Qualitative terms
- Concentrated solution
- Dilute solution
- To calculate the percent composition of a component in a compound:
- Find the molar mass of the compound by adding up the masses of each atom in the compound using the periodic table or a molecular mass calculator.
- Calculate the mass due to the component in the compound you are for which you are solving by adding up the mass of these atoms.
- Divide the mass due to the component by the total molar mass of the compound and multiply by 100.
- Mass percent( aka weight percent)
- Mass percent= (mass of solute/mass of solution) X100
You dilute a solution whenever you add solvent to a solution.
- Adding solvent results in a solution of lower concentration. You can calculate the concentration of a solution following a dilution by applying this equation:
MiVi = MfVf
where M is molarity, V is volume, and the subscripts i and f refer to the initial and final values.
Or by using the equation:
- CV=CV
Concentration and Molarity
- Molarity (M) is the concentration of a solution expressed as the number of moles of solute per liter of solution
- In order to calculate the molarity of a solution, you need to know the number of moles of solute and the total volume of the solution.
*Here is a calculation for the Molarity of a solution.
Step 1 Calculate the number of moles of NaOH that are in 20.0 grams.
Look up the atomic masses for the elements in NaOH from the Periodic Table. The atomic masses are found to be:
Na is 23.0
H is 1.0
O is 16.0
Plugging these values:
1 mol NaOH weighs 23.0 g + 16.0 g + 1.0 g = 40.0 g
So the number of moles in 20.0 g is:
moles NaOH = 20.0 g × 1 mol/40.0 g = 0.500 mol
Step 2 Determine the volume of solution in liters.
1 liter is 1000 cm3, so the volume of solution is: liters solution = 482 cm3 × 1 liter/1000 cm3 = 0.482 liter
Step 3 Determine the molarity of the solution.
Simply divide the number of moles by the volume of solution to get the molarity:
molarity = 0.500 mol / 0.482 liter
molarity = 1.04 mol/liter = 1.04 M
Answer
The molarity of a solution made by dissolving 20.0 g of NaOH to make a 482 cm3 solution is 1.04 M
This is an example of polar and non polar compounds and solutions.
Example:
How many millilieters of 5.5 M NaOH are needed to prepare 300 mL of 1.2 M NaOH?
Solution:
5.5 M x V1 = 1.2 M x 0.3 L
V1 = 1.2 M x 0.3 L / 5.5 M
V1 = 0.065 L
V1 = 65 mL
Concentration and Molarity
http://www.occc.edu/kmbailey/Chem1115Tutorials/Molarity.htm
* This website helps solve problems using concentration and molarity and gives an explanation.
http://www.youtube.com/watch?v=SXf9rDnVFao
*This video explains concentration and molarity problems. http://misterguch.brinkster.net/PRA003.pdf
*This is an activity to help with concentration and molarity.
Mass Percent
http://www.chem.tamu.edu/class/majors/tutorialnotefiles/percentcomp.htm
*This website helps explain and solve mass percent equations.http://www.yenka.com/activities/Percentage_Composition_by_Mass_-_Activity/?decorator=yenkaactivityprintable
*This is an activity that explains how to solve mass percent problems, and helps understand mass percent.
Solutions
http://chemistry.bd.psu.edu/jircitano/solution.html
*This website gives explanations on solutions and the quantities included in a solution.
http://www.youtube.com/watch?v=9h2f1Bjr0p4
*This is a crash course video that explains and describes solutions.
Stoichiometry
http://chemwiki.ucdavis.edu/Analytical_Chemistry/Chemical_Reactions/Stoichiometry_and_Balancing_Reactions#Variation_in_Stoichiometric_Equations
*This website explains and gives examples of stoichiometry problems.
http://www.youtube.com/watch?v=XnfATaoubzA
*This video further explains stoichiometry problems and how to solve them. http://www.glenoaks.edu/facultystaff/FacultyWebSites/SarahSimmons/Documents/NSC-130%20Stoichiometry%20Worksheet.pdf
*This is a worksheet that provides extra practice with stoichiometry problems.
Dilutions
http://www.youtube.com/watch?v=v6dnEp58mVk
*This video explains and describes how to solve a dilution problem.
http://phet-downloads.colorado.edu/files/activities/3511/molarity_dilution_lab.pdf
*This is an activity to help better explain dilutions.
Polar/Non Polar molecules
http://www.youtube.com/watch?v=PVL24HAesnc
*This video is a crash course on polar and non polar molecules, and this video explains the difference between them and also gives examples.
http://www.sophia.org/tutorials/polar-and-non-polar-molecules--2
*This is an activity that helps explain polar and nonpolar compounds and molecules.
Mass Percent
http://www.youtube.com/watch?v=kFpb9vBy2Lw
*This video explains how to calculate mass percent and gives examples of mass percent problems.
Concentration/Molarity
http://www.youtube.com/watch?v=h0cdLIfus8c
*This is a video that explains how to calculate concentration and molarity and how to identify problems finding concentration and molarity.
How many millilieters of 5.5 M NaOH are needed to prepare 300 mL of 1.2 M NaOH?
Solution:
5.5 M x V1 = 1.2 M x 0.3 L
V1 = 1.2 M x 0.3 L / 5.5 M
V1 = 0.065 L
V1 = 65 mL
Concentration and Molarity
http://www.occc.edu/kmbailey/Chem1115Tutorials/Molarity.htm
* This website helps solve problems using concentration and molarity and gives an explanation.
http://www.youtube.com/watch?v=SXf9rDnVFao
*This video explains concentration and molarity problems. http://misterguch.brinkster.net/PRA003.pdf
*This is an activity to help with concentration and molarity.
Mass Percent
http://www.chem.tamu.edu/class/majors/tutorialnotefiles/percentcomp.htm
*This website helps explain and solve mass percent equations.http://www.yenka.com/activities/Percentage_Composition_by_Mass_-_Activity/?decorator=yenkaactivityprintable
*This is an activity that explains how to solve mass percent problems, and helps understand mass percent.
Solutions
http://chemistry.bd.psu.edu/jircitano/solution.html
*This website gives explanations on solutions and the quantities included in a solution.
http://www.youtube.com/watch?v=9h2f1Bjr0p4
*This is a crash course video that explains and describes solutions.
Stoichiometry
http://chemwiki.ucdavis.edu/Analytical_Chemistry/Chemical_Reactions/Stoichiometry_and_Balancing_Reactions#Variation_in_Stoichiometric_Equations
*This website explains and gives examples of stoichiometry problems.
http://www.youtube.com/watch?v=XnfATaoubzA
*This video further explains stoichiometry problems and how to solve them. http://www.glenoaks.edu/facultystaff/FacultyWebSites/SarahSimmons/Documents/NSC-130%20Stoichiometry%20Worksheet.pdf
*This is a worksheet that provides extra practice with stoichiometry problems.
Dilutions
http://www.youtube.com/watch?v=v6dnEp58mVk
*This video explains and describes how to solve a dilution problem.
http://phet-downloads.colorado.edu/files/activities/3511/molarity_dilution_lab.pdf
*This is an activity to help better explain dilutions.
Polar/Non Polar molecules
http://www.youtube.com/watch?v=PVL24HAesnc
*This video is a crash course on polar and non polar molecules, and this video explains the difference between them and also gives examples.
http://www.sophia.org/tutorials/polar-and-non-polar-molecules--2
*This is an activity that helps explain polar and nonpolar compounds and molecules.
Mass Percent
http://www.youtube.com/watch?v=kFpb9vBy2Lw
*This video explains how to calculate mass percent and gives examples of mass percent problems.
Concentration/Molarity
http://www.youtube.com/watch?v=h0cdLIfus8c
*This is a video that explains how to calculate concentration and molarity and how to identify problems finding concentration and molarity.
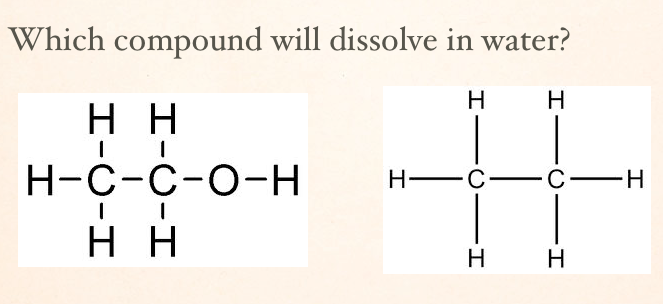
The compound that will dissolve in water will be the one with the OH group because it is more polar.
Mass Percent
- A solution is prepared by dissolving 1.0 g of sodium Chloride in to 48 g of water. What is the mass percent of sodium?
- Mass percent= Mass of solute/Mass of solution x100
- Mass percent= 1/49x100
- Mass Percent= 2.0%
This is a calculation using dilutions.
This is a calculation of stoichiometry problems and how to solve them.