Chapter 3
Matter
The stuff of which the universe is composed.
All matter has two characteristics:
States of Matter
-The distinct forms that different phases of matter take on
- Solid- rigid, has a fixed shape and volume; Ex. ICE
- Liquid- has a definite volume and takes the shape of the container; Ex. WATER
- Gas- Has no fixed volume or shape; takes the shape and volume of its container; Ex. Steam
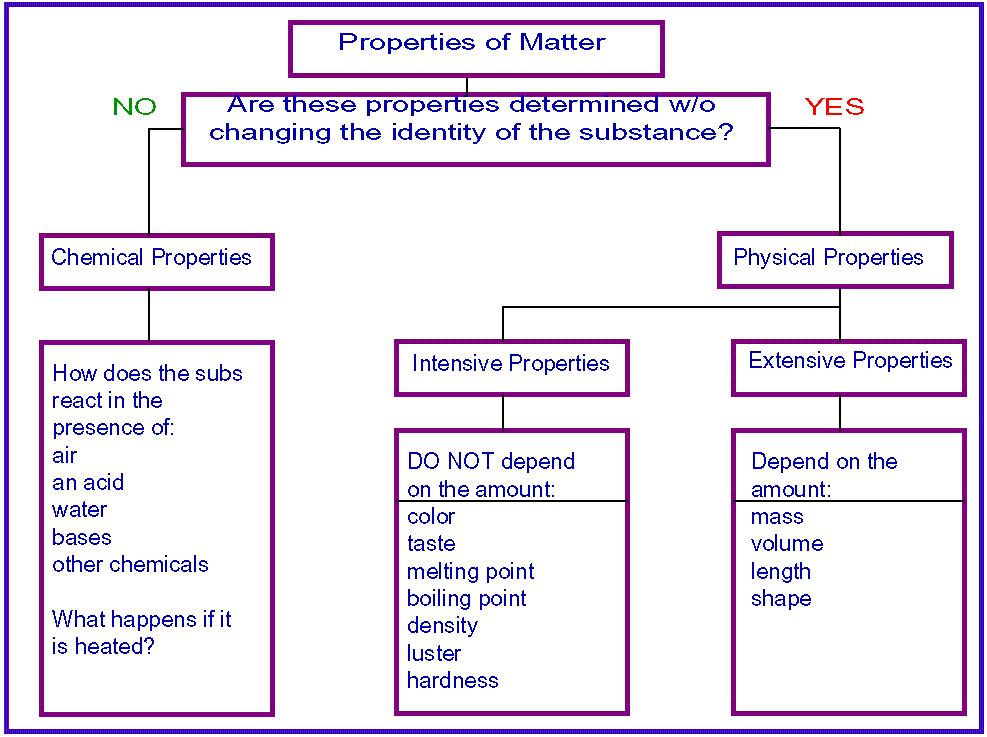
Physical Properties -
Are characteristics of a substance as it exist that can be observed and measurable.
Chemical Properties-
Physical Change/Chemical Change
Conservation of Mass
Atoms
Element
Protons
Neutrons
Electrons
Compounds
Substances
Mixtures
Separations of Mixtures
NEEDING EXTRA HELP?
Click on these links to take you to websites and videos where you can get extra help on these topics
Activities/Websites/Videos:
States of Matter
http://quizlet.com/25764087/states-of-matter-flash-cards/
*This activity helps better explain the different states of matter and gives examples of each.
http://www.chsd.us/~tthompson/assignments/Physical%20Science/work%20sheet%20physical%20chem%20changes.pdf
*This is an activity with the chemical and physical properties of matter.
http://www.teachengineering.org/view_activity.php?url=collection/uoh_/activities/uoh_sep_mixtures_activity1/uoh_sep_mixtures_activity1.xml
*This is an activity that explains the difference between elements mixtures and compounds.
http://www.proprofs.com/quiz-school/story.php?title=pure-substances-mixtures-quiz
* This is a quiz to take as a activity that quizzes on the topics of pure substances and mixtures. http://everythingscience.co.za/grade-10/02-classification-of-matter/02-classification-of-matter-02.cnxmlplus
* This is an activity to help with homogenous and heterogenous mixtures.
States of Matter
http://www.chem.purdue.edu/gchelp/atoms/states.html
http://www.youtube.com/watch?v=bMbmQzV-Ezs
*This video is on the states of matter that explains the three states of matter, gives examples of each state of matter, and also gives characteristic properties of each state of matter.
http://www.youtube.com/watch?v=bMbmQzV-Ezs
Properties of Matter
http://www.chem4kids.com/files/matter_intro.htmlhttp://schools.bcsd.com/fremont/5th_sci__matter_properties_of_matter.htm
*These are two websites that are useful in determining the properties of matter.
http://www.youtube.com/watch?v=C4pQQQNwy30
*This Video on the Properties of Matter explains the physical and chemical properties of matter and physical and chemical changes of matter.
http://studyjams.scholastic.com/studyjams/jams/science/matter/properties-of-matter.htm
*This is an activity to help with the properties of matter.
Physical Properties
http://www.elmhurst.edu/~chm/vchembook/104Aphysprop.html
*This website explains the physical properties of matter.
Intrinsic/Extrinsic Physical properties
http://www.youtube.com/watch?v=R2WV37PurRA
* This video explains intrinsic and extrinsic properties.
Chemical Properties
http://www.elmhurst.edu/~chm/vchembook/105Achemprop.html
*This website explains the chemical properties of matter.
Compounds
http://www.chem4kids.com/files/atom_compounds.htmlhttp://www.youtube.com/watch?v=VOQzWLeZoEc
* This Video explains elements and compounds and states the differences between them.
Mixtures/Pure Substances
http://chemsite.lsrhs.net/Intro/Pure_vs_mixtures.html
* This website gives examples and explains both mixtures and pure substances.
http://www.youtube.com/watch?v=M8sAYedRHO8
*This video explains the differences between mixtures and pure substances and gives examples of each of them.
Mixtures
http://www.chem4kids.com/files/matter_mixture.html
*This website describes the types of mixtures and it also explains the separations of mixtures.
Homogenous/Heterogenous Mixtures
http://www.youtube.com/watch?v=Wc9eTtAyrKc
* This video further explains the differences between homogenous and heterogenous mixtures.
Separating Mixtures
http://www.youtube.com/watch?v=F4x6OUUXoPs
*This video explains the ways to separate a mixture.
Matter
The stuff of which the universe is composed.
All matter has two characteristics:
- Mass
- Occupies space
States of Matter
-The distinct forms that different phases of matter take on
- Solid- rigid, has a fixed shape and volume; Ex. ICE
- Liquid- has a definite volume and takes the shape of the container; Ex. WATER
- Gas- Has no fixed volume or shape; takes the shape and volume of its container; Ex. Steam
Physical Properties -
Are characteristics of a substance as it exist that can be observed and measurable.
- Two type of physical properties:
- Intrinsic physical properties- a property of a system or of a material itself or within
- Ex. Density, Color, Odor, Hardness, Boiling Point, freezing point, states of matter and melting point
- Extrinsic physical properties- A property that is not essential or inherent
- Ex. Mass, volume, length, and dimension
Chemical Properties-
- The ability to form new substances under given conditions
- Octane( the major component of gasoline) + O2+ spark --> combustion( release of energy) + CO2 +H2O
Physical Change/Chemical Change
- Physical Change- A change that does not change the identity of the substances
- Chemical Change- A change that causes a substance to become a new substance or broken down into simpler substances
- All matter can go into Physical and chemical Change.
- Ice, water and Steam are made up of H2O
- The difference between the three states of matter is the spacing of the molecule and how ridged the spacing of the molecules
- By adding electricity to water, we can break water (H2O) to Hydrogen gas( H2) and Oxygen gas( O2).
- The decomposition of water by electricity is Electrolysis.
Conservation of Mass
- Matter cannot be created or destroyed but it can be converted into a different form.
Atoms
- Atoms is the smallest basic unit of matter.
Element
- A pure chemical substance consisting of one type of atom distinguished by its atomic number, which is the number of protons in its nucleus.
Protons
- Positive charge
Neutrons
- No charge
Electrons
- Negative charge
Compounds
- A substance made of atoms of different elements bonded together in a certain ratio
- One type of compound will always have the same combination of atoms.
Substances
- Most of matter around us consists of mixtures of substances.
- Example look at the soil. You can see different types of components like sand, clay, and remnants of plants
- Pure Substance: Materials that will always have the same composition
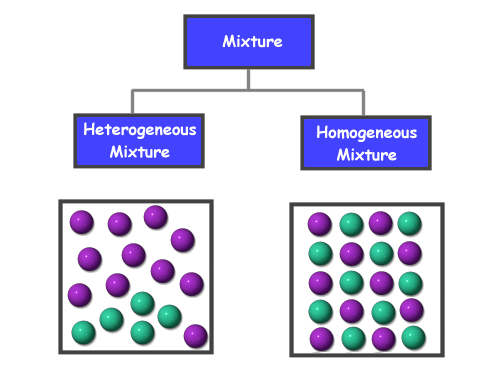
Mixtures
- Materials that has variable composition and is not chemically bonded
- Homogeneous Mixture- is the same proportion throughout the material
- Heterogenous Mixture- contains regions of different proportion
Separations of Mixtures
- Mixtures can be separated into pure substances either elements and/or compounds
- Distillation- The method that uses the idea heating solutions to separate the different liquids and some solids
- Filtration- separates liquid from a solid
- A liquid passes through the filter paper, but the particles are trapped
- In both separation method, there were no chemical changes that occurred.
- These methods are physical methods to purify the mixture
NEEDING EXTRA HELP?
Click on these links to take you to websites and videos where you can get extra help on these topics
Activities/Websites/Videos:
States of Matter
http://quizlet.com/25764087/states-of-matter-flash-cards/
*This activity helps better explain the different states of matter and gives examples of each.
http://www.chsd.us/~tthompson/assignments/Physical%20Science/work%20sheet%20physical%20chem%20changes.pdf
*This is an activity with the chemical and physical properties of matter.
http://www.teachengineering.org/view_activity.php?url=collection/uoh_/activities/uoh_sep_mixtures_activity1/uoh_sep_mixtures_activity1.xml
*This is an activity that explains the difference between elements mixtures and compounds.
http://www.proprofs.com/quiz-school/story.php?title=pure-substances-mixtures-quiz
* This is a quiz to take as a activity that quizzes on the topics of pure substances and mixtures. http://everythingscience.co.za/grade-10/02-classification-of-matter/02-classification-of-matter-02.cnxmlplus
* This is an activity to help with homogenous and heterogenous mixtures.
States of Matter
http://www.chem.purdue.edu/gchelp/atoms/states.html
http://www.youtube.com/watch?v=bMbmQzV-Ezs
*This video is on the states of matter that explains the three states of matter, gives examples of each state of matter, and also gives characteristic properties of each state of matter.
http://www.youtube.com/watch?v=bMbmQzV-Ezs
Properties of Matter
http://www.chem4kids.com/files/matter_intro.htmlhttp://schools.bcsd.com/fremont/5th_sci__matter_properties_of_matter.htm
*These are two websites that are useful in determining the properties of matter.
http://www.youtube.com/watch?v=C4pQQQNwy30
*This Video on the Properties of Matter explains the physical and chemical properties of matter and physical and chemical changes of matter.
http://studyjams.scholastic.com/studyjams/jams/science/matter/properties-of-matter.htm
*This is an activity to help with the properties of matter.
Physical Properties
http://www.elmhurst.edu/~chm/vchembook/104Aphysprop.html
*This website explains the physical properties of matter.
Intrinsic/Extrinsic Physical properties
http://www.youtube.com/watch?v=R2WV37PurRA
* This video explains intrinsic and extrinsic properties.
Chemical Properties
http://www.elmhurst.edu/~chm/vchembook/105Achemprop.html
*This website explains the chemical properties of matter.
Compounds
http://www.chem4kids.com/files/atom_compounds.htmlhttp://www.youtube.com/watch?v=VOQzWLeZoEc
* This Video explains elements and compounds and states the differences between them.
Mixtures/Pure Substances
http://chemsite.lsrhs.net/Intro/Pure_vs_mixtures.html
* This website gives examples and explains both mixtures and pure substances.
http://www.youtube.com/watch?v=M8sAYedRHO8
*This video explains the differences between mixtures and pure substances and gives examples of each of them.
Mixtures
http://www.chem4kids.com/files/matter_mixture.html
*This website describes the types of mixtures and it also explains the separations of mixtures.
Homogenous/Heterogenous Mixtures
http://www.youtube.com/watch?v=Wc9eTtAyrKc
* This video further explains the differences between homogenous and heterogenous mixtures.
Separating Mixtures
http://www.youtube.com/watch?v=F4x6OUUXoPs
*This video explains the ways to separate a mixture.
This image explains the physical and chemical properties of matter and gives examples of intrinsic extrinsic properties as well.
Intrinsic/Extrinsic Properties
Physical/Chemical Properties
- Height of a person- intrinsic or extrinsic
- Odor after a workout-intrinsic or extrinsic
Physical/Chemical Properties
- Octane( the major component of gasoline) + O2+ spark --> combustion( release of energy) + CO2 +H2O
- A. The boiling point of a certain alcohol is 78 degree Celsius
- B. Diamond is very hard
This is an example of elements and compounds and this shows the differences between both elements and compounds and the one similarity they share in common.
Compounds/Elements
Compounds/Elements
- Compounds- are made up of elements combined together in a fixed ratio.
- Elements- are found on the periodic table
- Is H2O a compound or an element?
This is an example of the comparison of both mixtures and pure substances and what makes up both of them.
Mixtures/Pure Substance
Mixtures/Pure Substance
- Mixtures- Defined as materials that have variable composition and are not chemically bonded.
- Ex. Coffee, Salad, Dirt
- Pure Substances- are materials that will always have the same composition
- Ex. Pure H20, CO, CO2
This shows an example of both homogenous and heterogenous mixtures and the makeup of both types of mixtures.
Homogenous/Heterogeneous Mixtures
Homogenous/Heterogeneous Mixtures
- Homogenous mixtures- have the same proportion throughout the material
- Ex. Air, Gasoline
- Heterogeneous Mixtures- contains regions of different proportion
- Ex. Fruit Salad, Dirt, Stream with gravel at the bottom