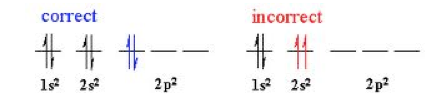Chapter 11: Modern Atomic Theory
Main Concept: Electromagnetic Radiation
http://www.lbl.gov/MicroWorlds/ALSTool/EMSpec/
This video explains electromagnetic radiation:
- Energy is being transmitted from one place to another by light
- Different types
- Wavelength - the distance between two consecutive wave peaks
- Frequency - indicates how many wave peaks pass a certain point per given time period
- Light travels in waves
- Photons - a stream of tiny packets of energy that create a light beam by traveling through space
- Different wavelengths of electromagnetic radiation carry different amounts of energy
- The photons that correspond to red light carry less energy that the photons that correspond to blue light
- The longer the wavelength, the lower the energy of its photons
http://www.lbl.gov/MicroWorlds/ALSTool/EMSpec/
This video explains electromagnetic radiation:
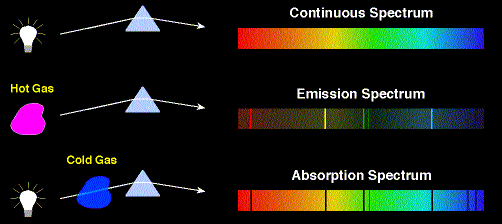
Main Concept: Emissions by Atoms
- When atoms receive energy from a source they become excited and they can release this energy by emitting light
- The emitted energy is carried away by a photon
- The energy of the photon corresponds exactly to the energy change experienced by the emitting atom
- High-energy photons correspond to short-wavelength light and low-energy photons correspond to long-wavelength light
- The photons of red light carry less energy than the photons of blue light because red light has a longer wavelength than blue light does
Main Concept: Energy levels of Atoms
- An atom with excess energy is said to be in an excited state
- An excited atom can release some of all of its excess energy by emitting a photon and then moves to a lower energy state
- The lowest possible energy state of an atom is called its ground state
- Energy contained in the photon corresponds to the change in energy that the atom experiences in going from the excited state to the lower state
- Only certain energy levels can occur
Main Concept: Difference between Continuous and Discreet Energy Levels
Quantized energy levels result from the relation between a particle's energy and its wavelength. For a confined particle such as an electron in an atom, the wave function has the form of standing waves. Only stationary states with energies corresponding to integral numbers of wavelengths can exist; for other states the waves interfere destructively, resulting in zero probability density (http://en.wikipedia.org/wiki/Energy_level)
Quantized energy levels result from the relation between a particle's energy and its wavelength. For a confined particle such as an electron in an atom, the wave function has the form of standing waves. Only stationary states with energies corresponding to integral numbers of wavelengths can exist; for other states the waves interfere destructively, resulting in zero probability density (http://en.wikipedia.org/wiki/Energy_level)
Main Concept: Wave Mechanical Model of the Atom
- This model gives no information about when the electron occupies a certain point in space of how it moves
- There are reasons to believe that we can never know the details of electron motion
- The electron does not orbit the nucleus in circles
Main Concept: Different Types of Molecular Orbitals
http://www.science.uwaterloo.ca/~cchieh/cact/c120/mo.html
- Although the probability of finding the electron decreases at greater distances from the nucleus, the probability of finding it even at great distances from the nucleus never becomes exactly zero
http://www.science.uwaterloo.ca/~cchieh/cact/c120/mo.html
Main Concept: Pauli Exclusion Principal
- States that an atomic orbital can only have a maximum of two electrons
- The electrons have to have opposite spins
- The spin of electrons are represented with arrows
This video explains the Pauli Exclusion Principal
Main Point: Electron Configuration
The electron configuration of an atom is the representation of the arrangement of electrons that are distributed among the orbital shells and subshells
This Website explains electron configuration:
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Electronic_Configurations
This Activity has problems with electron configuration:
http://www.learner.org/interactives/periodic/elementary_interactive.html
This is a Calculation of electron configuration:
The electron configuration of an atom is the representation of the arrangement of electrons that are distributed among the orbital shells and subshells
This Website explains electron configuration:
http://chemwiki.ucdavis.edu/Inorganic_Chemistry/Electronic_Configurations
This Activity has problems with electron configuration:
http://www.learner.org/interactives/periodic/elementary_interactive.html
This is a Calculation of electron configuration:
This video explains electron configuration
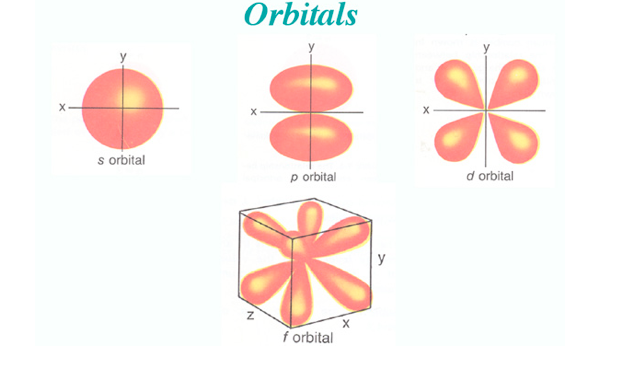
Main Point: Orbital Diagram
orbital: probability map for electrons
This Website explains orbital diagrams:
http://en.wikibooks.org/wiki/High_School_Chemistry/Orbital_Configurations
This Activity has problems with orbital diagrams:
http://www.lcps.org/cms/lib4/VA01000195/Centricity/ModuleInstance/74890/Orbital%20Diagrams%20Activity.pdf
This is an orbital diagram Calculation:
orbital: probability map for electrons
This Website explains orbital diagrams:
http://en.wikibooks.org/wiki/High_School_Chemistry/Orbital_Configurations
This Activity has problems with orbital diagrams:
http://www.lcps.org/cms/lib4/VA01000195/Centricity/ModuleInstance/74890/Orbital%20Diagrams%20Activity.pdf
This is an orbital diagram Calculation:
This video explains orbital diagrams
Main Point: Valence and Core Electrons
This Activity has problems with valence and core electrons
http://chemwiki.ucdavis.edu/Wikitexts/UC_Davis/UCD_Chem_124A%3A_Kauzlarich/ChemWiki_Module_Topics/Valence_and_Core_Electrons
- Valence Electrons - the electrons in the outermost principal energy level of an atom
- Core Electrons - the inner electrons and are not involved in bonding atoms to each other
This Activity has problems with valence and core electrons
http://chemwiki.ucdavis.edu/Wikitexts/UC_Davis/UCD_Chem_124A%3A_Kauzlarich/ChemWiki_Module_Topics/Valence_and_Core_Electrons
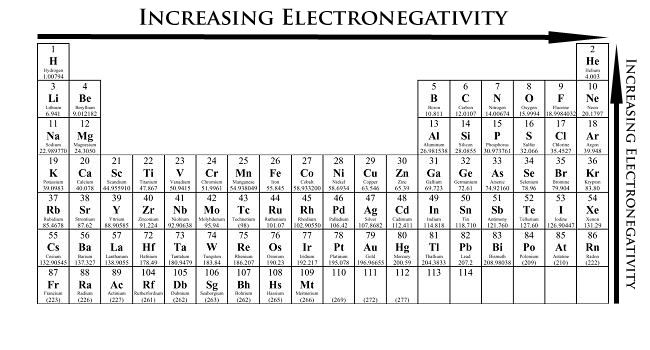
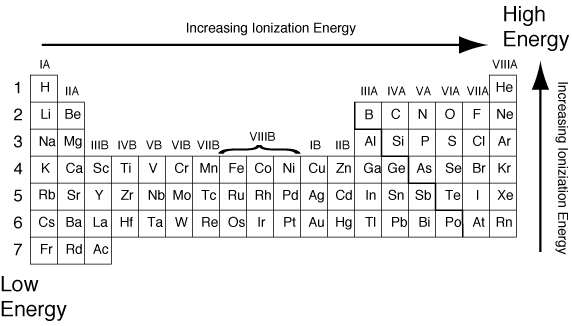
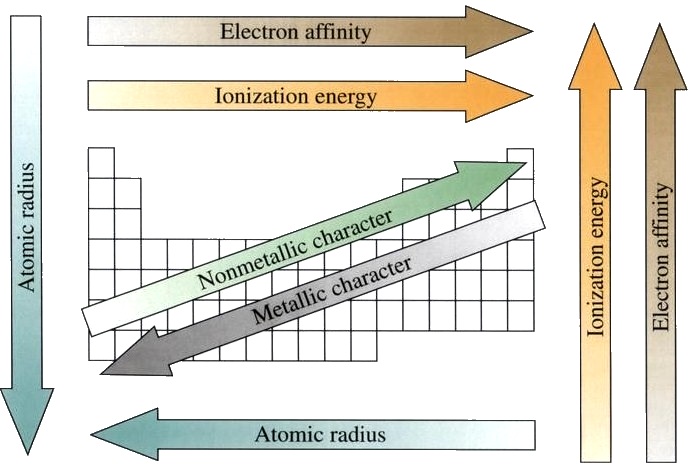
Main Point: Atomic Properties, the Periodic Table, and Periodic Table Trends
This Activity has problems with atomic properties, the periodic table, and the periodic table trends:
http://www.learner.org/interactives/periodic/periodic_table.html
This is an example Calculation of an atomic property:
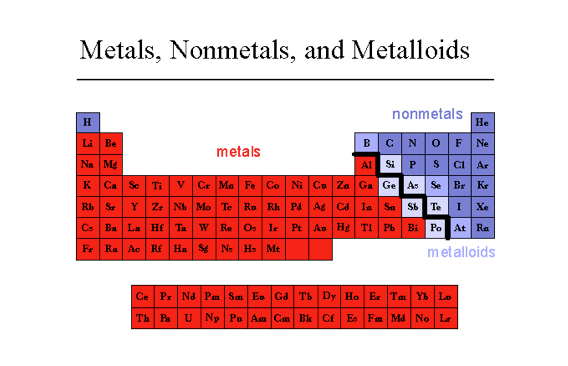
- Metals - lustrous/change shape/conduct heat and electricity
- Non-Metals - no physical properties like metals
- Metalloids - have metallic and nonmetallic physical properties
This Activity has problems with atomic properties, the periodic table, and the periodic table trends:
http://www.learner.org/interactives/periodic/periodic_table.html
This is an example Calculation of an atomic property:
This is a video that explains periodic table tren