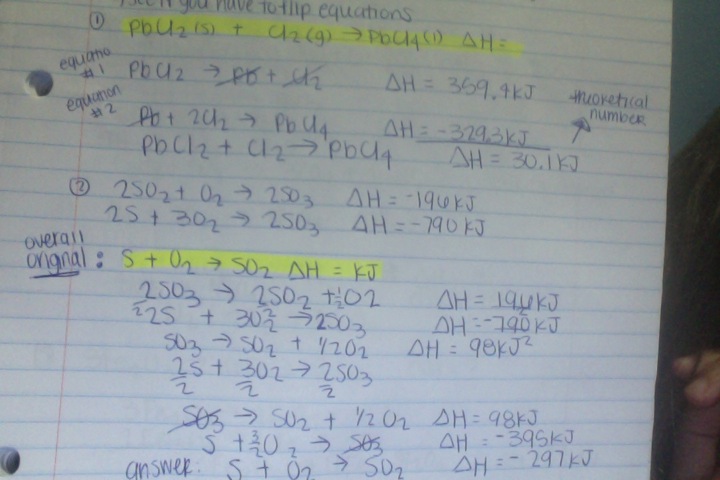Main Points:
What is energy?
-ability to do work
-ability to produce heat
Website Link: This website goes into detail of the definition of energy.
Law of Conservation of Energy
-can't be created
-can't be destroyed
-CAN be converted from one form to another
work= force times distance
-in order to work, you must move
-based on the effects around the environment
Website Link: This website further explains what the Law of Conservation of Energy is.
Temperature and Heat
What is Temperature?
-measure of the random motions of the components of a substance
What is heat?
- flow of energy due to a temperature difference
-high to low
-high specific heat are good insulators
-low specific heat are good conductors
Website Link: This website goes into detail in the explanation of temperature and heat.
Exothermic and Endothermic:
endothermic- absorbs heat
exothermic- gives off heat
Website Link: This website explains what exothermic and endothermic is in detail.
Video Link: This video goes in depth of the differences between exothermic and endothermic.
Thermodynamics:
thermodynamics- the study of how energy flows
Website Link: This website goes into detail of the topic of thermodynamics.
Specific Heat Capacity:
Heat capacity: the measurable physical quantity of heat energy required to change the temperature of an object by a given amount.
internal energy- change in energy= q+w (change in energy= heat+work)
exothermic-= change in energy= -q + w
units of heat= joules= J + Calories=- C
1Cal= 4.184J
formulas:
q= MC(change in temperature)
q measures HEAT of a system
w measures WORK of a system
Video Link: This video further explains how to calculate the specific heat capacity.
Activity Link: This activity gives problems involving specific heat capacity and helps reiterate the understanding of this topic.
Website Link: This website goes into detail of how to configure the specific heat capacity.
Calculations: When you have a problem with the equation q=mc(Tf-Ti), first list out what you have, then rearrange the equation, and then plug in the numbers.
q=-9750J
m= 335g
c= 4.184J/g®C
Tf= ??
Ti= 65.5®C
To solve, use the equation below:
q=mc(Tf-Ti)
q/mc=Tf-Ti
q/mc+Ti= Tf
-9750J/335g*4.184J/g®C+65.5®C
Answer: 58.5
Enthalpy:
Enthalpy- difference between reaction and product
product up higher= increase endothermic
product of bottom= decrease- exothermic
Website Link: This website further explains what Enthalpy is and helps one better understand the concept.
Video Link: This video goes into detail about what Enthalpy is.
Hess's Law:
Hess's Law: The law states that the total enthalpy change during the complete course of a reaction is the same whether the reaction is made in one step or in several steps.
Hess's law steps
1. see which one you need to flip if you do. when you flip it change the sign of the ΔH that goes with it.
2. multiply and/or divide to make the coefficients equal
3. cancel out
4. check to make sure it looks like the overall equation
5. add/subtract the ΔH's
Website Link: This website goes into detail about Hess's Law.
Activity Link: This activity gives problems that contain the concept of Hess's Law and helps one better understand this topic and how to calculate the problems containing this subject.
Video Link: This video goes in depth of the explanation for Hess's Law.
Calculations:
Use the following data to determine the enthalpy
(ΔH°) of reaction for: Overall: PCl5(g)->PCl3(g)+Cl2(g)ΔH=?
1)P4(s)+6Cl2(g)->4PCl3(g)
ΔH=-2439kJ
2)4PCl5(g)->P4(s)+10Cl2(g)Δ=3438kJ
Answer=250. kJ
Energy as a Driving Force:
Calculating Energy:
Activity Link: This website contains the explanation of how to calculate energy and includes an activity to help practice the understanding of how to calculate energy.
Video Link: This video goes in depth of explaining how to calculate energy.
Calculations:
-ability to do work
-ability to produce heat
Website Link: This website goes into detail of the definition of energy.
Law of Conservation of Energy
-can't be created
-can't be destroyed
-CAN be converted from one form to another
work= force times distance
-in order to work, you must move
-based on the effects around the environment
Website Link: This website further explains what the Law of Conservation of Energy is.
Temperature and Heat
What is Temperature?
-measure of the random motions of the components of a substance
What is heat?
- flow of energy due to a temperature difference
-high to low
-high specific heat are good insulators
-low specific heat are good conductors
Website Link: This website goes into detail in the explanation of temperature and heat.
Exothermic and Endothermic:
endothermic- absorbs heat
exothermic- gives off heat
Website Link: This website explains what exothermic and endothermic is in detail.
Video Link: This video goes in depth of the differences between exothermic and endothermic.
Thermodynamics:
thermodynamics- the study of how energy flows
Website Link: This website goes into detail of the topic of thermodynamics.
Specific Heat Capacity:
Heat capacity: the measurable physical quantity of heat energy required to change the temperature of an object by a given amount.
internal energy- change in energy= q+w (change in energy= heat+work)
exothermic-= change in energy= -q + w
units of heat= joules= J + Calories=- C
1Cal= 4.184J
formulas:
q= MC(change in temperature)
q measures HEAT of a system
w measures WORK of a system
Video Link: This video further explains how to calculate the specific heat capacity.
Activity Link: This activity gives problems involving specific heat capacity and helps reiterate the understanding of this topic.
Website Link: This website goes into detail of how to configure the specific heat capacity.
Calculations: When you have a problem with the equation q=mc(Tf-Ti), first list out what you have, then rearrange the equation, and then plug in the numbers.
q=-9750J
m= 335g
c= 4.184J/g®C
Tf= ??
Ti= 65.5®C
To solve, use the equation below:
q=mc(Tf-Ti)
q/mc=Tf-Ti
q/mc+Ti= Tf
-9750J/335g*4.184J/g®C+65.5®C
Answer: 58.5
Enthalpy:
Enthalpy- difference between reaction and product
product up higher= increase endothermic
product of bottom= decrease- exothermic
Website Link: This website further explains what Enthalpy is and helps one better understand the concept.
Video Link: This video goes into detail about what Enthalpy is.
Hess's Law:
Hess's Law: The law states that the total enthalpy change during the complete course of a reaction is the same whether the reaction is made in one step or in several steps.
Hess's law steps
1. see which one you need to flip if you do. when you flip it change the sign of the ΔH that goes with it.
2. multiply and/or divide to make the coefficients equal
3. cancel out
4. check to make sure it looks like the overall equation
5. add/subtract the ΔH's
Website Link: This website goes into detail about Hess's Law.
Activity Link: This activity gives problems that contain the concept of Hess's Law and helps one better understand this topic and how to calculate the problems containing this subject.
Video Link: This video goes in depth of the explanation for Hess's Law.
Calculations:
Use the following data to determine the enthalpy
(ΔH°) of reaction for: Overall: PCl5(g)->PCl3(g)+Cl2(g)ΔH=?
1)P4(s)+6Cl2(g)->4PCl3(g)
ΔH=-2439kJ
2)4PCl5(g)->P4(s)+10Cl2(g)Δ=3438kJ
Answer=250. kJ
Energy as a Driving Force:
- energy spread - in a given process, concentrated energy is dispersed widely
- matter spread - the molecules of a substance are spread out and occupy a larger volume
- entropy ( S ) - a measure of disorder and randomness
- second law of thermodynamics: The entropy of the universe is always increasing.
- spontaneous process - process that occurs in nature without outside intervention; it happens "on its own"
Calculating Energy:
Activity Link: This website contains the explanation of how to calculate energy and includes an activity to help practice the understanding of how to calculate energy.
Video Link: This video goes in depth of explaining how to calculate energy.
Calculations: